The ACES® clinical study1
The ACES Study was led by investigators from the Interventional Pulmonology Research Center at Vanderbilt. Other sites included Johns Hopkins, University of North Carolina, and Memorial Health System in Florida. The study enrolled 25 patients with aseptic pleural effusions that had recurred after a previous thoracentesis. The primary endpoint was the percentage change in pleural effusion volume on chest CT from baseline to 30 days.
Patient Population
Patients who received ACES included individuals with pleural effusions related to lung cancer, breast cancer, and renal failure, as well as those with idiopathic pleural effusions. Clinical outcomes were evaluated for the first 20 patients at 30 days and for the first 17 patients at 60 days.
Significant effusion reduction1
The graphic below summarizes changes in effusion volumes within the treated hemithorax at 30 days (n=23) and 60 days (n=20), illustrating the progression of fluid reduction and lung re-expansion over time. Up to a 94% reduction in median pleural effusion volume was observed.
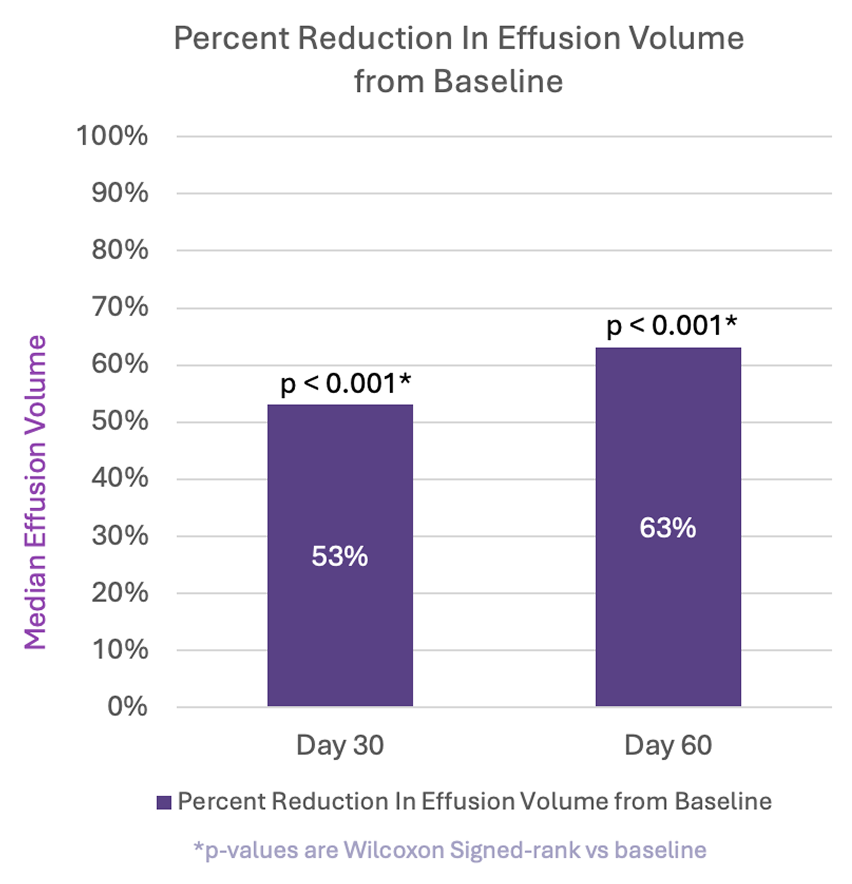
Up to 94% reduction in pleural effusion volume
Fluid clearance maintained through 60 days1
In a study patient, fluid occupying nearly half of the hemithorax at baseline was significantly reduced by 30 days and remained low at 60 days, demonstrating consistent drainage and lung re-expansion over time.
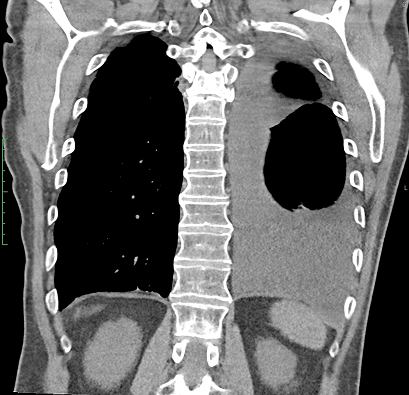
CT Baseline
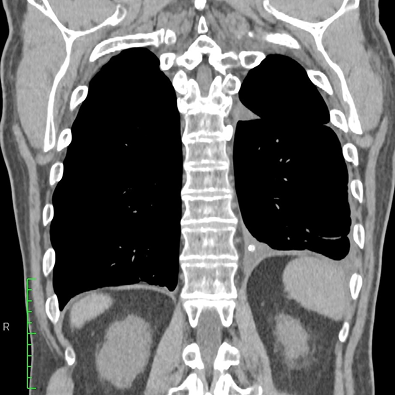
30 Days
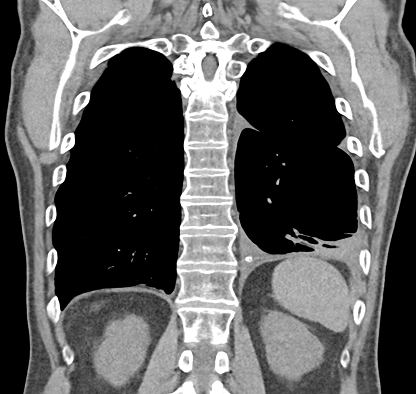
60 Days
Meaningful safety profile1

n=25
- Zero pleural infections
- Zero reported ascites or tumor seeding in clinical study

“The device works. It goes in, stays in, drains fluid, doesn’t clog, patients feel better, they don’t need bottles, and they don’t need to come in for management… this thing is doing what it's supposed to do.”
—Lonny Yarmus, DO, Baltimore, MD
Low risk of abdominal tumor seeding1-8
- Average life expectancy for patients with malignant pleural effusions is 4.5 months2
- Cancer cell seeding and growth times much longer3,4
- No incidence of seeding or ascites in ACES study patients1
- Clinically detectable tumor spread to abdomen with Denver Shunt exceedingly low and restricted to patients with malignant pleural mesothelioma5,6
- Peritoneal spread of abdominal and lung cancers is rare7,8
The risk of clinically meaningful spread of cancer cells to the abdomen following placement of a pleuroperitoneal shunt is extremely low
References:
1. Data on File. Pleural Dynamics. The ACES Study for Aseptic Pleural Effusion. ClinicalTrials.gov ID NCT06210685. 2. Clive AO, Kahan BC, Hooper CE, et al. Predicting survival in malignant pleural effusion: development and validation of the LENT prognostic score. Thorax. 2014;69(12):1098-1104. 3. Harris K, Khachaturova I, Azab B, et al. Small cell lung cancer doubling time and its effect on clinical presentation: a concise review. Clin Med Insights Oncol. 2012;6:199-203. 4. Dahan M, Hequet D, Bonneau C, Paoletti X, Rouzier R. Has tumor doubling time in breast cancer changed over the past 80 years? A systematic review. Cancer Med. 2021;10(15):5203-5217. 5. Genc O, Petrou M, Ladas G, Goldstraw P. The long-term morbidity of pleuroperitoneal shunts in the management of recurrent malignant effusions. Eur J Cardiothorac Surg. 2000;18(2):143-146. 6. Collins DC, Sundar R, Constantinidou A, et al. Radiological evaluation of malignant pleural mesothelioma—defining distant metastatic disease. BMC Cancer. 2020;20(1):1210. 7. Tuthill M, Pell R, Guiliani R, et al. Peritoneal disease in breast cancer: a specific entity with an extremely poor prognosis. Eur J Cancer. 2009;45(12):2146-2149. 8. Satoh H, Ishikawa H, Yamashita YT, et al. Peritoneal carcinomatosis in lung cancer patients. Oncol Rep. 2001;8(6):1305-1307.
The ACES implant is approved for sale in the USA. Caution: Federal (United States) law restricts this device to sale by or on order of a physician. Prior to use, please see the Instructions for Use for a complete listing of Indications, Contraindications, Warnings, Precautions, Potential Complications, and Insertion Instructions.

